Recent Post

The Difference Between Pull Force, Holding Force, and Shear Force – A Common Misconception

Magnet components – the unsung hero of the green transition

How do you choose the perfect magnetic strength for your application?
The effects of the conflict in the Middle East

Quality Assurance of Magnets
Is gold and silver magnetic materials?
Published: 2023-04-20 16:45:40 • Daniel Gårdefelt
The world of magnetism is very interesting and different materials have different magnetic properties. Gold and silver, two of the most valuable precious metals, have been used for many things, from jewelry to money, for hundreds of years. But are gold and silver attracted to magnets? In this article we will talk about how magnetic these metals are and how they work with magnets.
Diamagnetism, Paramagnetism and Ferromagnetism
To understand how gold and silver are magnetic, you need to know the three main types of magnetism: diamagnetism, paramagnetism, and ferromagnetism.
Diamagnetic materials have a weak, negative (opposite pole) sensitivity to magnetic fields. Diamagnetic materials are easily repelled by a magnetic field and do not retain the magnetic properties when the external field is removed. In diamagnetic materials, all the electrons are paired so that there is no permanent net magnetic moment per atom. Diamagnetic properties arise from the rearrangement of electron orbits under the influence of an external magnetic field. Most elements in the periodic table, including copper, silver, and gold, are diamagnetic.
Paramagnetic materials have a small positive sensitivity to magnetic fields. These materials are somewhat attracted by a magnetic field but, as with diamagnetic materials, do not retain the magnetic properties when the external field is removed. Paramagnetic properties depend on the presence of some unpaired electrons and on the rearrangement of electron orbits caused by the external magnetic field. Paramagnetic materials include magnesium, molybdenum, lithium and tantalum.
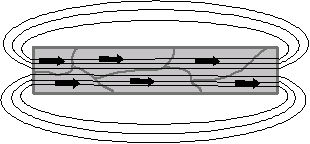
Ferromagnetic materials have a large, positive sensitivity to an external magnetic field. They exhibit a strong attraction to magnetic fields and can retain their magnetic properties after the external field is removed. Ferromagnetic materials have some unpaired electrons so their atoms have a net magnetic moment. They get their strong magnetic properties due to the presence of magnetic domains. In these domains, a large number of atomic moments (1012 to 1015) are aligned in parallel so that the magnetic force within the domain is strong. When a ferromagnetic material is in the unmagnetized state, the domains are almost randomly organized and the net magnetic field of the part as a whole is zero. When a magnetizing force is applied, the domains become aligned to produce a strong magnetic field within the part. Iron, nickel and cobalt are examples of ferromagnetic materials. Components with these materials are usually inspected using the magnetic particle method
 |
 |
The magnetic properties of gold and silver
Both gold and silver are diamagnetic, meaning they have a weak, negative (opposite) susceptibility to magnetic fields.
In short, magnets do not attract gold and silver as they do ferromagnetic materials such as iron, nickel and cobalt. Gold and silver are diamagnetic, meaning they are slightly repelled by a magnetic field.


